Accueil / Information / Aller plus loin... / Aériens / Environnement intérieur / Les acariens
Les acariens
dimanche 8 juillet 2012, par ,
Les acariens sont des arthropodes. Le schéma ci-dessous donne un aperçu de la phylogénèse des acariens, ainsi que de l’extrême diversité de leur écologie :
La pathologie due aux acariens n’est pas limitée aux réactions allergiques respiratoires :
- affections cutanées : gale
 , demodex, ..
, demodex, .. - morsures de tiques




- réactions alimentaires dues à des farines contaminées
Elle affecte aussi de nombreux animaux de compagnie et d’élevage (mammifères, oiseaux). Qui, à leur tour, peuvent entraîner une allergie chez l’homme ![]() .
.
Les acariens phytophages et les acariens prédateurs utilisés contre ces derniers sont également la cause d’allergies respiratoires professionnelles ![]()
![]()
![]()
![]() .
.
D’innombrables études ont été menées pour comprendre l’épidémiologie, la pathogénie ou la nature des allergènes des acariens. Elles se sont concentrées sur un nombre relativement restreint d’espèces, au premier rang desquelles Dermatophagoides farinae, Dermatophagoides pteronyssinus et Blomia tropicalis.
Ces acariens dits « domestiques » sont une cause majeure d’allergie respiratoire persistante. Leurs caractéristiques allergologiques sont détaillées ci-après, en même temps que celles d’autres espèces d’acariens retrouvés dans les habitations ou dans des environnements favorables à leur croissance (acariens dits de stockage).
Des réactivités croisées ont été décrites entre acariens et d’autres invertébrés, posant la question d’une association pertinente ou non sur le plan clinique : se reporter aux thèmes crustacés, mollusques marins, escargots et blattes.
Les allergènes des acariens
On ne connaît pas moins de 100 allergènes différents ayant pour origine les acariens ![]()
![]()
![]() . Ce grand nombre s’explique par :
. Ce grand nombre s’explique par :
- la fréquence de l’allergie aux acariens et le caractère mondial de celle-ci
- la variété des espèces d’acariens
- la richesse en molécules IgE-réactives différentes révélées avec les acariens quand on les étudie avec des méthodes séparatives, comme les immuno-empreintes.
La plupart des allergènes d’acariens ont reçu une dénomination officielle (IUIS).
Le tableau suivant donne un résumé des allergènes (et/ou protéines IgE-réactives) caractérisés à ce jour :
L’usage est resté de parler de « groupes », chaque groupe correspondant, en fait, à des allergènes de même fonction biochimique. Quand cette fonction n’est pas encore connue, on classera dans un même groupe des molécules suffisamment homologues les unes des autres (ex. groupe 5).
Contrairement à d’autres produits allergisants, par exemple les poissons, les acariens n’ont pas un allergène vraiment dominant : la majeure partie de l’IgE-réactivité est répartie entre 4-5 groupes au moins.
La relevance clinique n’a pas été contrôlée pour chacun des allergènes des acariens, notamment par des tests de provocation nasale et/ou bronchique. Parmi les allergènes d’acariens, beaucoup sont plutôt des « molécules IgE-réactives ».
Les propriétés des différents groupes ![]()
![]()
![]()
![]()
![]()
![]()
![]()
- Groupe 1







 : les allergènes du groupe 1 sont des cystéine protéases, c’est-à-dire des enzymes capables de scinder d’autres protéines. Ce sont les premiers allergènes d’acariens à avoir été caractérisés : Der p 1 en 1980
: les allergènes du groupe 1 sont des cystéine protéases, c’est-à-dire des enzymes capables de scinder d’autres protéines. Ce sont les premiers allergènes d’acariens à avoir été caractérisés : Der p 1 en 1980  et Der f 1 en 1982
et Der f 1 en 1982  . Der p 1 et Der f 1 ont une homologie de séquence de 85%. Si l’homologie est forte entre Der p 1, Der f 1 et Eur m 1 (Euroglyphus), elle tombe à moins de 35% entre ces allergènes et Blo t 1 (Blomia), et plus encore en surface des protéines
. Der p 1 et Der f 1 ont une homologie de séquence de 85%. Si l’homologie est forte entre Der p 1, Der f 1 et Eur m 1 (Euroglyphus), elle tombe à moins de 35% entre ces allergènes et Blo t 1 (Blomia), et plus encore en surface des protéines  , de sorte que Blo t 1 ne croise pas avec le groupe 1 des acariens Pyroglyphides.
, de sorte que Blo t 1 ne croise pas avec le groupe 1 des acariens Pyroglyphides.
La masse moléculaire de Der p 1 est de 25 kDa
A noter l’absence de groupe 1 (pour le moment) dans les acariens « de stockage » type Lepidoglyphus.
Les allergènes du groupe 1 sont principalement dans les feces des acariens ![]()
![]() . Ils représentent une part significative de la réactivité dans un extrait d’acarien Dermatophagoides, même si, malgré une tendance à la standardisation des extraits, des concentrations assez différentes sont relevées d’un fournisseur à un autre.
. Ils représentent une part significative de la réactivité dans un extrait d’acarien Dermatophagoides, même si, malgré une tendance à la standardisation des extraits, des concentrations assez différentes sont relevées d’un fournisseur à un autre.
Le groupe 1 des acariens appartient aux cystéine protéases C1. Ces enzymes sont dites aussi « papaïne-like ». On ne s’étonnera pas de trouver des homologues dans la papaye (papaïne), dans l’ananas (ananaïne, broméline), dans le soja (Gly m Bd30K), etc.. Si des réactions croisées avec ces cystéines protéases végétales ont lieu ![]()
![]() , elles sont très marginales. Il faut dire que les pourcentages d’identité entre ces protéines ne sont pas favorables à une réactivité croisée.
, elles sont très marginales. Il faut dire que les pourcentages d’identité entre ces protéines ne sont pas favorables à une réactivité croisée.
L’impact de Der p 1 sur l’immunité ne se limite pas à une IgE réactivité. Sa fonction protéase le rend capable de capable de modifier la perméabilité de la barrière bronchique épithéliale. Il est également susceptible d’induire la libération de médiateurs pro-inflammatoires et la production de récepteurs de chimiokines. Der p 1 joue aussi un rôle direct sur les mastocytes, les éosinophiles, les cellules musculaires lisses bronchiques ![]() .
.
Les allergènes du groupe 1 comportent tous un site de N-glycolysation au niveau des résidus 53-55, ce qui explique la présence d’hydrates de carbone dans l’extrait naturel purifié. En outre, en conditions physiologiques, l’existence d’une structure non glycolysée sous forme d’un dimère de 40 kDa a été mis en évidence ![]() .
.
- Groupe 2













 : ces protéines font partie d’une grande famille possédant un domaine dit « ML domain lipid binding ». Elles ont une cavité apte à transporter un stéroïde, par exemple. On les trouve dans les feces, mais moins électivement que pour le groupe 1. Elles seraient secrétées par les acariens mâles et semblent particulièrement résistantes à la chaleur.
: ces protéines font partie d’une grande famille possédant un domaine dit « ML domain lipid binding ». Elles ont une cavité apte à transporter un stéroïde, par exemple. On les trouve dans les feces, mais moins électivement que pour le groupe 1. Elles seraient secrétées par les acariens mâles et semblent particulièrement résistantes à la chaleur.
Les allergènes du groupe 2 ont quelque homologie avec MD2, une molécule du complexe Toll TLR-4. A ce titre, elles pourraient jouer un rôle immuno-modulateur.
L’homologie est faible entre Der p 2 / f 2 et Blo t 2 (Blomia) ou les allergènes d’acariens « de stockage » comme Gly d 2 (Glycyphagus), Lep d 2 (Lepidoglyphus), Tyr p 2 (Tyrophagus). Chez ces derniers, faute de groupe 1, le groupe 2 tient une place importante.
Les allergènes du groupe 2 ont une masse moléculaire de 14 à 18 kDa.
Les allergènes du groupe 2 se distinguent par la présence de lignages différents selon les régions du monde, ainsi que par des isoformes qui ne suivent pas la taxonomie.
- Groupe 3






 : ce sont des sérine protéases comme pour les groupes 6 et 9. Mais le groupe 3 (trypsine-like) a moins de 40% d’identité avec le groupe 6 (chymotrypsine-like) ou avec le groupe 9 (collagénase-like). Peu d’homologie également avec la trypsine bovine (env. 40% d’identité).
: ce sont des sérine protéases comme pour les groupes 6 et 9. Mais le groupe 3 (trypsine-like) a moins de 40% d’identité avec le groupe 6 (chymotrypsine-like) ou avec le groupe 9 (collagénase-like). Peu d’homologie également avec la trypsine bovine (env. 40% d’identité).
Ces allergènes sont trouvés dans les feces. Ils présentent un très grand polymorphisme. Leur allergénicité serait importante pour certains auteurs, mais leur fragilité dans les extraits tend à expliquer la faible IgE-réactivité constatée pour ces allergènes.
Le poids moléculaire des allergènes du groupe 3 est de 30 kDa.
Les allergènes du groupe 3 sont capables de cliver le complément avec pour conséquence la production des anaphylatoxines C3a et C5a ![]() .
.
- Groupe 4

 : ce sont des alpha amylases. Elles présentent une meilleure homologie entre Der p 4 / f 4 et Blo t 4 (env. 65% d’identité) qu’il n’est vu pour d’autres groupes.
: ce sont des alpha amylases. Elles présentent une meilleure homologie entre Der p 4 / f 4 et Blo t 4 (env. 65% d’identité) qu’il n’est vu pour d’autres groupes.
Der p 4 a un poids moléculaire de 15 kDa.
- Groupe 5






 : bien que d’origine digestive ces allergènes ne sont pas que dans les feces. Leur fonction biochimique est inconnue pour le moment. Ils comprennent des isoformes très dissemblables (env 35% d’identité) et ne croisant pas entre elles.
: bien que d’origine digestive ces allergènes ne sont pas que dans les feces. Leur fonction biochimique est inconnue pour le moment. Ils comprennent des isoformes très dissemblables (env 35% d’identité) et ne croisant pas entre elles.
Blo t 5 est l’allergène le plus important de Blomia tropicalis. Il ne croise pas avec Der p 5 / f 5. L’identité de séquence de Blo t 5 est de 43% avec Der p 5, de 40% avec Der p 21 alors que ce dernier n’a qu’une identité de séquence de 30% avec Der p 5.
Der p 5 a une masse moléculaire de 15 kDa.
- Groupe 6 : ils ne croisent pas avec le groupe 3 (il existe une faible identité de séquence entre les deux groupes) et leur part dans l’IgE-réactivité globale semble très faible.
Der p 6 et Der f 6 ont un poids moléculaire de 25 kDa.
- Groupe 7


 : ces allergènes n’ont pas de fonction biochimique caractérisée pour le moment. Ils sont fragiles et se retrouvent sous formes de fragments très divers dans les extraits. De la sorte, l’évaluation exacte de leur allergénicité est difficile. Certains auteurs pensant qu’elle pourrait être aussi importante que celle du groupe 1.
: ces allergènes n’ont pas de fonction biochimique caractérisée pour le moment. Ils sont fragiles et se retrouvent sous formes de fragments très divers dans les extraits. De la sorte, l’évaluation exacte de leur allergénicité est difficile. Certains auteurs pensant qu’elle pourrait être aussi importante que celle du groupe 1.
Les allergènes du groupe 7 ont un poids moléculaire de 22 kDa.
- Groupe 8


 : ce sont des glutathion S-transférases (GST). Ces enzymes sont ubiquitaires au sein du monde vivant. Les GST ne sont pas dans les feces et leur IgE-réactivité est très mineure.
: ce sont des glutathion S-transférases (GST). Ces enzymes sont ubiquitaires au sein du monde vivant. Les GST ne sont pas dans les feces et leur IgE-réactivité est très mineure.
Les acariens possèdent des GST de classe µ, laquelle a moins de 30% d’identité avec les GST des plantes ou des insectes ![]() . Une réactivité croisée des GST d’acariens avec celle d’autres GST est donc peu probable. Une étude a cependant noté une inhibition de Der p 8 par la blatte américaine chez 2 patients/6
. Une réactivité croisée des GST d’acariens avec celle d’autres GST est donc peu probable. Une étude a cependant noté une inhibition de Der p 8 par la blatte américaine chez 2 patients/6 ![]() .
.
Der p 8 est un polypeptide d’un poids moléculaire de 27 kDa.
- Groupe 9 : ce sont des sérine protéases à activité collagénase. Une homologie avec des sérine protéases de crustacés a été avancée
 mais calculée que sur une courte séquence N-terminale. Comme Der p 3, Der p 9 est susceptible d’induire la libération de GM-CSF et d’éotaxine en activant le récepteur PAR-2 des cellules épithéliales et de cliver les jonctions serrées ce qui entraîne une augmentation de la perméabilité de l’épithélium bronchique
mais calculée que sur une courte séquence N-terminale. Comme Der p 3, Der p 9 est susceptible d’induire la libération de GM-CSF et d’éotaxine en activant le récepteur PAR-2 des cellules épithéliales et de cliver les jonctions serrées ce qui entraîne une augmentation de la perméabilité de l’épithélium bronchique  .
.
Der p 9 a un poids moléculaire de 28 kDa.
- Groupe 10

 : les tropomyosines des acariens sont suspectées de générer des réactions croisées avec celles d’autres arthropodes comme les crustacés ou les blattes. La pertinence clinique de ces RC est abordée ailleurs (Crustacés, Blattes, Mollusques et escargots).
: les tropomyosines des acariens sont suspectées de générer des réactions croisées avec celles d’autres arthropodes comme les crustacés ou les blattes. La pertinence clinique de ces RC est abordée ailleurs (Crustacés, Blattes, Mollusques et escargots).
Les Helminthes sont également à l’origine d’une IgE-réactivité vis-à-vis des tropomyosines et peuvent perturber l’interprétation des résultats pour les acariens. Une réactivation d’allergie à l’Anisakis chez un patient au cours de l’immunothérapie pour les acariens a même été rapportée ![]() .
.
En Europe, la prévalence de positivité pour le groupe 10 est plus faible que dans des régions d’endémie parasitaire.
Le poids moléculaire de Der p 10 est de 36 kDa.
- Groupe 11





 : ces protéines de forte masse (env. 100 kDa) sont des paramyosines. Comme les tropomyosines elles sont d’origine musculaire. Leur instabilité génère des formes dégradées, de sorte qu’il est difficile d’estimer l’importance clinique de ces allergènes.
: ces protéines de forte masse (env. 100 kDa) sont des paramyosines. Comme les tropomyosines elles sont d’origine musculaire. Leur instabilité génère des formes dégradées, de sorte qu’il est difficile d’estimer l’importance clinique de ces allergènes.
On trouve des paramyosines dans certains parasites (Anisakis, Taenia, Schistosoma). Les pourcentages d’identité étant faibles (souvent 30-35% d’identité), les réactions croisées des paramyosines d’acariens avec celles de ces parasites semblent peu probables.
Les protéines du groupe 11 ont un poids moléculaire élevé de 92-94 kDa.
- Groupe 12 : ce sont des protéines capables de se lier à la chitine. Les allergènes du groupe 12 de L. destructor et B. tropicalis ont une forte identié de séquence de 93%.
Ce groupe, à la fonction inconnue, n’a pas été décrit pour les acariens Pyroglyphides. Par contre, ils ont été retrouvé chez les acariens de stockage et Blomia tropicalis
Blo t 12 a un poids moléculaire de 14 kDa pour une séquence de 124 acides aminés.
- Groupe 13


 : ces protéines peuvent transporter des molécules lipophiles comme les acides gras. Bien qu’en concentration notable, leur IgE-réactivité semble très limitée.
: ces protéines peuvent transporter des molécules lipophiles comme les acides gras. Bien qu’en concentration notable, leur IgE-réactivité semble très limitée.
Le recombinant rBlo t 13 a un poids moléculaire de 14,8 kDa.
- Groupe 14








 : ces protéines appartiennent à un ensemble dénommé « large LTP » qui contient notamment des vitellogénines de crustacés et des apolipophorines d’insectes. Elles sont habituellement désignées comme des apolipophorines. Ce sont des molécules de forte masse, plus ou moins hydrosolubles et mal représentées dans les extraits. Instables, elles génèrent des fragments (ex. M177 qui a 177 kDa). Mais ceux-ci conservent une IgE-réactivité notable.
: ces protéines appartiennent à un ensemble dénommé « large LTP » qui contient notamment des vitellogénines de crustacés et des apolipophorines d’insectes. Elles sont habituellement désignées comme des apolipophorines. Ce sont des molécules de forte masse, plus ou moins hydrosolubles et mal représentées dans les extraits. Instables, elles génèrent des fragments (ex. M177 qui a 177 kDa). Mais ceux-ci conservent une IgE-réactivité notable.
Une certaine confusion entoure les dénominations du groupe 14 : « Mag-3 » est un recombinant issu de D. farinae qui a servi (comme « Mag-1 ») à générer des anticorps utilisés pour repérer des allergènes homologues dans d’autres acariens.
Beaucoup d’auteurs considèrent le groupe 14 comme important cliniquement.
- Groupe 15


 : ces chitinases de fort poids moléculaire (env. 100 kDa) se distinguent par leur allergénicité de premier ordre chez le chien et le chat mais apparemment pas chez l’homme. Présentes dans l’appareil digestif des acariens, elles ne sont pas en quantités notables dans les feces. Ces protéines sont fortement O-glycosylées.
: ces chitinases de fort poids moléculaire (env. 100 kDa) se distinguent par leur allergénicité de premier ordre chez le chien et le chat mais apparemment pas chez l’homme. Présentes dans l’appareil digestif des acariens, elles ne sont pas en quantités notables dans les feces. Ces protéines sont fortement O-glycosylées.
Les chitinases sont connues comme étant le support des réactions alimentaires dans le syndrome « latex- fruits exotiques ». Cependant les protéines du groupe 15 des acariens sont des molécules très différentes des chitinases végétales. Pas de réaction croisée décrite à ce jour entre la banane et un acarien !
- Groupe 17
 : ce sont des protéines possédant un site dit « EF-hand » capable de lier le calcium (comme les parvalbumines). IgE-réactivité faible.
: ce sont des protéines possédant un site dit « EF-hand » capable de lier le calcium (comme les parvalbumines). IgE-réactivité faible.
- Groupe 18

 : comme les chitinases du groupe 15, on ne les retrouve pas dans les feces. Ces chitinases sont particulières car leur domaine C-terminal, qui devrait porter l’activité enzymatique, est tronqué. Le groupe 18 ne croise pas avec le groupe 15 et a une IgE-réactivité faible.
: comme les chitinases du groupe 15, on ne les retrouve pas dans les feces. Ces chitinases sont particulières car leur domaine C-terminal, qui devrait porter l’activité enzymatique, est tronqué. Le groupe 18 ne croise pas avec le groupe 15 et a une IgE-réactivité faible.
Ces chitinases jouent un rôle dans l’inflammation et le remodelage tissulaire des mammifères.
Der f 18 a un poids moléculaire de 60 kDa.
- Groupe 19 : on ne connaît que Blo t 19. Et encore : cet allergène adoubé par l’IUIS n’a pas été publié. Il aurait une vague homologie avec un « antimicrobial factor » d’Ascaris.
Cet allergène de B. tropicalis a une masse moléculaire de 7 kDa.
- Groupe 20


 : ce sont des arginine kinases. Ces enzymes suivent de près les tropomyosines : on les retrouve également chez les crustacés et les insectes. Elles semblent avoir une IgE-réactivité très limitée, notamment par leur faible concentration.
: ce sont des arginine kinases. Ces enzymes suivent de près les tropomyosines : on les retrouve également chez les crustacés et les insectes. Elles semblent avoir une IgE-réactivité très limitée, notamment par leur faible concentration.
Les protéines du groupe 20 ont un poids moléculaire de 40 kDa.
- Groupe 21

 : on ne connaît pas la fonction biochimique de ces protéines si ce n’est qu’elles ont une petite homologie avec celles du groupe 5 (mais ne croisent pas avec elles). Elles ont une origine intestinale et semblent bien résister à la chaleur.
: on ne connaît pas la fonction biochimique de ces protéines si ce n’est qu’elles ont une petite homologie avec celles du groupe 5 (mais ne croisent pas avec elles). Elles ont une origine intestinale et semblent bien résister à la chaleur.
- Groupe 22
 : on ne connaît pour l’instant que Der f 22. Cet allergène est d’origine intestinale, a 35% d’identité avec Der f 2 et a été trouvé IgE-réactif chez 50% des patients.
: on ne connaît pour l’instant que Der f 22. Cet allergène est d’origine intestinale, a 35% d’identité avec Der f 2 et a été trouvé IgE-réactif chez 50% des patients.
- Allergènes non encore rangés dans un groupe :
- Les alpha tubulines


 : ce sont des protéines ubiquitaires trouvées dans le cytosquelette des cellules. Celle de Lepidoglyphus a été montrée IgE-réactive chez 12% de patients suédois, et celle de Tyroglyphus chez 29% de coréens
: ce sont des protéines ubiquitaires trouvées dans le cytosquelette des cellules. Celle de Lepidoglyphus a été montrée IgE-réactive chez 12% de patients suédois, et celle de Tyroglyphus chez 29% de coréens - Les thaumatine-like

 : ces protéines sont connues comme allergènes dans les pollens de Cupressacées et dans certains fruits (pomme, cerise, kiwi). Des résultats préliminaires semblent montrer la possibilité d’une réactivité croisée entre acariens et végétaux par le truchement de ces thaumatine-like : l’acarien Glycyphagus domesticus possède une thaumatine-like qui présente 62% d’identité avec Mal d 2, la thaumatine-like de la pomme. Or, si l’on mesure la fréquence de positivité pour la thaumatine-like de Glycyphagus, on trouve 12% chez des sujets allergiques aux acariens (Singapour), mais pas moins de 78% chez des polliniques (Italie) ! A moins d’une interférence par des CCD, on pourrait donc se trouver en présence d’une réactivité croisée végétal/animal.
: ces protéines sont connues comme allergènes dans les pollens de Cupressacées et dans certains fruits (pomme, cerise, kiwi). Des résultats préliminaires semblent montrer la possibilité d’une réactivité croisée entre acariens et végétaux par le truchement de ces thaumatine-like : l’acarien Glycyphagus domesticus possède une thaumatine-like qui présente 62% d’identité avec Mal d 2, la thaumatine-like de la pomme. Or, si l’on mesure la fréquence de positivité pour la thaumatine-like de Glycyphagus, on trouve 12% chez des sujets allergiques aux acariens (Singapour), mais pas moins de 78% chez des polliniques (Italie) ! A moins d’une interférence par des CCD, on pourrait donc se trouver en présence d’une réactivité croisée végétal/animal. - les heat shock proteins (HSP) sont des protéines ubiquitaires : les moisissures et la noisette, par exemple, ont des allergènes de ce type. La réactivité croisée avec les HSP d’acariens mériterait d’être étudiée
- Les alpha tubulines
Malgré le nombre élevé des allergènes (ou molécules IgE-réactives) déjà caractérisés chez les acariens, certains travaux semblent montrer que ce nombre est encore très en dessous de la réalité.
Ainsi, l’équipe de F.T. Chew, à Singapour, a notablement agrandie la panoplie des allergènes (ex. dans le cas de Blomia tropicalis) ou des protéines se présentant comme de sérieux candidats à l’IgE-réactivité.
- Ces auteurs ont décrit plus de 20 allergènes potentiels, jusque-là ignorés, dans Tyrophagus
 , dans Aleuroglyphus
, dans Aleuroglyphus  et dans Suidasia
et dans Suidasia  .
. - C’est ainsi que des familles d’allergènes décrites chez les végétaux, les moisissures, les mammifères pourraient trouver des membres parmi les acariens : des profilines, des cyclophilines, des thioredoxines, etc..
- En produisant des recombinants pour certaines de ces protéines, Chew a pu montrer leur IgE-réactivité. Il a notamment caractérisé 2 homologues d’allergènes d’œuf de poule : une ovalbumine dans Tyrophagus, Suidasia et D. farinae ; et un ovomucoïde dans Blomia. Ces protéines sont mêmes assez fréquemment positives (in vitro) chez des patients allergiques aux acariens (Singapour)
 .
.
- Est-ce que les ovalbumine et/ou ovomucoïde d’œuf de poule croisent avec ces homologues d’acariens ? Cela ne semble pas avoir été étudié jusqu’alors. La question est d’intérêt quand on sait que la positivité pour l’œuf à 6 mois/1 an est un assez bon facteur prédictif pour une allergie respiratoire à 4-7 ans.
Activité biochimique et allergénicité
Parallèlement à leur IgE-réactivité, les extraits d’acariens semblent posséder des propriétés biochimiques favorisant leur allergénicité.
Il s’agit avant tout de l’activité protéasique des allergènes du groupe 1 (Der p 1, Der f 1) qui a été étudiée et, à un moindre degré, celle des sérine protéases (groupes 3 et 9 ![]() ). Il faut remarquer que d’autres protéines dans les extraits (allergènes non identifiés, etc…) pourraient contribuer aussi bien à cet effet adjuvant (et à sa compensation) et donc que ces travaux ne sont pas le reflet fidèle du contact réel avec les poussières d’acariens.
). Il faut remarquer que d’autres protéines dans les extraits (allergènes non identifiés, etc…) pourraient contribuer aussi bien à cet effet adjuvant (et à sa compensation) et donc que ces travaux ne sont pas le reflet fidèle du contact réel avec les poussières d’acariens.
Globalement, l’activité protéasique serait pro-inflammatoire et favoriserait les passages trans-épithéliaux des allergènes :
- modification des molécules CD40, CD25, CD23
 , DC-SIGN
, DC-SIGN  , PAR2
, PAR2 
- attaque des surfactants A et D
 et des inhibiteurs de protéases
et des inhibiteurs de protéases 

- effet direct sur les cellules épithéliales

 et leurs jonctions
et leurs jonctions 
- stimulation de la réponse des cellules dendritiques


Malgré tous ces résultats in vitro ou chez l’animal ![]()
![]()
![]() , des questions restent en suspends : ces expériences sont-elles réalistes (concentrations d’allergènes utilisées) ? Les conditions expérimentales sont-elles transposables à l’homme ? Certains experts sont sceptiques
, des questions restent en suspends : ces expériences sont-elles réalistes (concentrations d’allergènes utilisées) ? Les conditions expérimentales sont-elles transposables à l’homme ? Certains experts sont sceptiques ![]()
![]() . D’autant que chez le chat et le chien les allergènes dominants (groupe15) n’ont pas d’activité protéasique.
. D’autant que chez le chat et le chien les allergènes dominants (groupe15) n’ont pas d’activité protéasique.
Acariens : polyréactivité / monoréactivité
Réagit-on aux acariens de Métropole quand on arrive d’Outre-Mer ? Et inversement. Est-il important de tester les 2 Dermatophagoides ? L’environnement professionnel est-il à prendre en compte en cas d’allergie due à des acariens domestiques ?
Les réponses à ces questions sont tributaires de plusieurs paramètres intriqués et néanmoins indépendants les uns des autres :
L’homologie entre allergènes d’acariens différents :
Les divergences entre protéines s’élèvent avec l’éloignement taxonomique des espèces (cf. le schéma taxonomique).
En termes de possibilités de réactivité croisée, plus l’identité entre 2 protéines est proche de 100% et plus on a des chances de rencontrer des réactions croisées entre ces protéines.
Le tableau suivant donne une idée des divergences entre allergènes d’acariens :
| Par rapport à | D. Farinae | Blomia tropicalis | Lepidoglyphus destructor | Tyrophagus putrescenciae | |
|---|---|---|---|---|---|
| D. Pteronyssinus | Groupe 1 | 82 % d’identité | 34 % | ||
| Groupe 2 | 88 % | 39 % | 36 % | 27 % | |
| Groupe 3 | 81 % | 48 % | |||
| Groupe 4 | 90 % | 68 % | |||
| Groupe 5 | 42 % | 30 % | |||
| Groupe 6 | 75 % | 58 % | |||
| Groupe 7 | 86 % | 29 % | |||
| Groupe 9 | 72 % | 56 % | |||
| Groupe 10 | 98 % | 94 % | 94 % | 94 % | |
| Groupe 11 | 89 % | 78 % | |||
| D Farinae | Groupe 13 | 80 % | 79 % | 57 % |
On voit que des réactions croisées sont en théorie possibles entre allergènes du groupe 10 (tropomyosines) et peut-être au sein du groupe 13 (FABP) ; et aussi, bien sûr entre allergènes d’espèces proches comme D. farinae et D. pteronyssinus.
Mais ceci est malgré tout une simplification de la réalité.
De nombreux allergènes d’acariens se présentent sous plusieurs isoformes :
- par exemple, Der p 1 n’a pas une séquence unique d’acides aminés mais plusieurs variantes très proches les unes des autres (23 exactement, de Der p 1.0101 à Der p 1.0123).
- Ces isoformes sont même parfois en « désordre » par rapport à l’arbre taxonomique : par exemple Gly d 2 a une isoforme plus proche de Tyr p 2 que de Lep d 2
 .
.
Enfin, on peut trouver aussi des paralogues, c’est-à-dire des allergènes similaires mais ayant dérivé beaucoup plus tôt au cours de l’évolution de sorte que leurs pourcentages d’identité sont très faibles, de l’ordre de 30-40% :
- c’est le cas pour des allergènes du groupe 5 comme Der p 5 et Blo t 5
 ou Sui m 5
ou Sui m 5  .
. - Ces allergènes paralogues ne croisent pas entre eux.
L’ensemble de ces nuances constitue un éventail très large avec des proximités et des éloignements à la fois entre espèces et au sein des différents groupes d’allergènes.
Le résultat visible, à savoir la positivité des tests et les manifestations cliniques, sera la somme de toutes ces nuances. Il existe non pas une allergie aux acariens mais des allergies aux acariens.
L’environnement du patient, actuel mais aussi passé en cas de changement de lieu de vie, est un facteur majeur à prendre en considération pour comprendre une réactivité à plusieurs acariens.
Certains acariens ont été décrits principalement (ou uniquement) dans des régions à climat tropical ou sub-tropical :
- les Blomia : Canaries, Caraïbes, Amérique du Sud, Asie du Sud-Est, Taïwan
- les Suidasia : Malaisie, Singapour
- Dermatophagoides siboney : Cuba, Porto-Rico
On a retrouvé des acariens Dermatophagoides dans toutes les régions du monde. D. farinae et D. pteronyssinus sont généralement présents simultanément. Parfois D. farinae est majoritaire (ex. en Grande Bretagne, Australie, Nouvelle-Zélande, Corée, Thaïlande), parfois minoritaire (ex. Colombie, Cuba).
La répartition d’une même espèce d’acarien peut varier dans un même pays d’une région à une autre, en rapport avec l’humidité ambiante ![]() .
.
Les acariens domestiques se distinguent le plus souvent des acariens trouvés dans les lieux de stockage ou les bâtiments agricoles. Cependant, la frontière n’est pas absolue et Tyrophagus, par exemple, est un acarien domestique en Corée (cf. Acariens domestiques ou de stockage ?).
Le classique travail de M. Van Hage ![]() montre bien l’importance du répertoire local des acariens sur la réactivité observée :
montre bien l’importance du répertoire local des acariens sur la réactivité observée :
- ces auteurs ont comparé des patients Suédois exposés à Lepidoglyphus avec des patients Brésiliens exposés à Blomia tropicalis :
- In vitro, les patients étaient positifs pour les 2 acariens.
- Mais si Lepidoglyphus parvenait à inhiber le RAST Blomia chez les sujets Suédois, cette même réaction croisée n’était pas obtenue avec le sérum des patients Brésiliens.
- C’était même l’inverse : Blomia inhibait Lepidoglyphus avec le sérum des brésiliens et pas avec le sérum des suédois !
- Clairement, cela montrait que les IgE des suédois étaient beaucoup plus affines pour les épitopes de l’acarien local (Lepidoglyphus) que d’un acarien non présent dans l’environnement (Blomia). Et vice versa.
Des résultats similaires ont été montrés pour D. farinae et Blomia entre Singapour et la Thaïlande ![]() .
.
On voit qu’une positivité à plus d’un acarien peut recouvrir une véritable sensibilisation à un acarien et une réactivité croisée pour un autre, celle-ci pouvant être sans relevance clinique si la réactivité croisée est de faible affinité.
Les réactivités croisées entre allergènes d’acariens
Les limites de réactivité croisée sont assez parallèles aux pourcentages d’identité des allergènes deux à deux.
Ceci étant, ce sont surtout les modifications d’acides aminés situés en surface externe des allergènes qui vont limiter les possibilités de réactivité croisée ![]()
![]() .
.
Les allergènes les plus homologues entre les acariens peuvent constituer l’explication d’une réactivité croisée minimale, observée même si les acariens ne sont pas dans l’environnement du patient. On pense, bien sûr, avant tout aux tropomyosines ![]() ; mais les paramyosines (groupe 11) et les FABP (groupe 13) sont des candidats aussi.
; mais les paramyosines (groupe 11) et les FABP (groupe 13) sont des candidats aussi.
D’autres groupes d’allergènes sont peu propices à des réactions croisées, du moins entre les genres principaux que sont Blomia et Dermatophagoides. C’est le cas des groupes1, 2 et 5.
Mais si, de fait, Tyr p 2 (Tyrophagus, acarien de stockage) ne croise pas avec Der p 2, il croise très bien avec Lep d 2 et Gly d 2 qui lui sont plus proches taxonomiquement.
Globalement, on pourrait résumer ainsi :
- Les Dermatophagoides (farinae, pteronyssinus, et aussi siboney aux Caraïbes) croisent très aisément entre eux.
- Quelques cas de mono-réactivité à D. farinae sans réactivité à D. pteronyssinus (ou l’inverse) ont bien été décrits : ils restent très rares et soulignent, une fois encore, la possible variété des réponses immunologiques des patients face à un même allergène.
Une étude menée sur un collectif de 1500 patients français a montré seulement 16 cas de CAP positifs pour D. farinae ayant un résultat <0,35 kU/l pour D. pteronyssinus (et uniquement 2 résultats D. farinae positifs avec D. pteronyssinus <0,10 kU/l) (Laurence Guilloux, Biomnis, in press).
- Quelques cas de mono-réactivité à D. farinae sans réactivité à D. pteronyssinus (ou l’inverse) ont bien été décrits : ils restent très rares et soulignent, une fois encore, la possible variété des réponses immunologiques des patients face à un même allergène.
- Blomia tropicalis croise mal avec les Dermatophagoides.
- Une double positivité pour Blomia et Dermatophagoides dans un environnement favorable à Blomia représente donc a priori une double sensibilisation : d’une part le sujet s’est sensibilisé à Blomia et, d’autre part, il s’est indépendamment sensibilisé à Dermatophagoides. Cela peut être important sur le plan de l’immunothérapie.
- On a parfois trouvé des résultats positifs pour des tests in vitro Blomia dans des pays au climat incompatible avec Blomia tropicalis
 : cela semble du à la présence d’un autre Blomia, Blomia tjibodas, qui se satisfait de conditions climatiques plus froides
: cela semble du à la présence d’un autre Blomia, Blomia tjibodas, qui se satisfait de conditions climatiques plus froides  .
.
- Euroglyphus maynei est considéré comme un acarien domestique, cohabitant avec les Dermatophagoides.
- En général Euroglyphus est inhibé par D. farinae ou D. pteronyssinus, mais l’inverse est plus rare.
- Euroglyphus aurait quelques épitopes qui lui seraient propres. Mais la part prise par Euroglyphus sur le plan clinique reste floue.
- Dans certaines conditions d’exposition, Lepidoglyphus peut constituer l’acarien majeur pour les patients.
- Il a alors été obtenu des inhibitions partielles de Dermatophagoides par Lepidoglyphus, mais au prix de doses élevées de Lepidoglyphus (parfois 10000 fois plus que la dose homologue)
 .
. - Lepidoglyphus forme avec Tyrophagus et Glycyphagus un groupe volontiers cross-réactif.
- Acarus siro est souvent à part, croisant de manière inconstante avec les 3 précédents.
- Il en est de même pour Chortoglyphus. Tous ces acariens sont dits « de stockage ».
- La positivité pour un acarien de stockage chez un sujet non exposé professionnellement pose la question d’une réactivité croisée ou d’une réelle sensibilisation.
Les réactivités croisées entre acariens pourraient être résumées dans le schéma ci-dessous établi à partir du cumul de 24 études différentes, sachant que les disparités d’exposition sont bien sûr gommées (climat, pays, etc..).
- Il a alors été obtenu des inhibitions partielles de Dermatophagoides par Lepidoglyphus, mais au prix de doses élevées de Lepidoglyphus (parfois 10000 fois plus que la dose homologue)
Les chiffres représentent le nombre d’études où une réaction croisée a été observée (au moins chez la moitié des patients) sur le nombre total d’études. Par exemple, 5 fois /9 un Dermatophagoides inhibait Lepidoglyphus, alors que l’inhibition inverse n’était relevée qu’une fois/11.
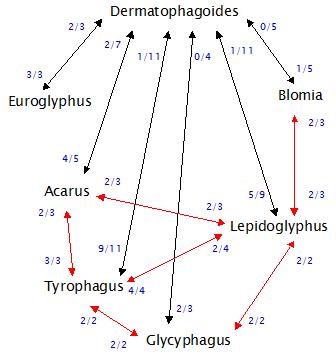
On remarquera que la réactivité croisée est en règle générale en faveur des acariens Dermatophagoides ; et que les RC entre acariens « de stockage » sont plus équilibrées.
La primauté des Dermatophagoides peut trouver plusieurs explications :
- il est rare que ces acariens n’accompagnent pas une exposition à un autre acarien. Par exemple, dans une étude menée à Huelva (Espagne) si des Glycyphagus étaient présents dans 54% des domiciles testés, on trouvait D. pteronyssinus dans 95% des domiciles

- par ailleurs, l’allergénicité des Dermatophagoides pourrait être dominante : dans la même étude, sur les 26 patients avec CAP positif pour Glycyphagus, seuls 3 avaient un CAP D. pteronyssinus nettement plus faible que celui pour Glycyphagus
Ceci étant, la réactivité croisée entre ces 2 acariens était quasi-absente et l’on peut dans le cas d’espèce pencher pour une double sensibilisation Glycyphagus + Dermatophagoides plutôt qu’une simple positivité pour Glycyphagus générée par des IgE anti-Dermatophagoides ![]() .
.
Acariens domestiques ou de stockage ?
Il est classique de distinguer les acariens de poussière de maison (« house dust mites ») des acariens que l’on rencontre volontiers dans des lieux à usage professionnel : comme ces lieux servent souvent de lieux d’entreposage, on a dénommé ces acariens « de stockage » (« storage mites »).
Cependant la distinction ne doit pas être considérée comme absolue et, dans des conditions favorables, tout acarien « de stockage » pourrait devenir un acarien de maison = un acarien domestique ![]() . C’est le cas pour Tyrophagus
. C’est le cas pour Tyrophagus ![]() , pour Chortoglyphus
, pour Chortoglyphus ![]() , pour Glycyphagus
, pour Glycyphagus ![]() , etc …
, etc …
Les lieux favorisant le développement de ces acariens sont les silos, les stocks d’aliments pour bétail, les meuneries, voire les boulangeries ou les animaleries ![]() . Les fermiers sont exposés notamment là où le travail auprès des animaux s’effectue pendant une longue période hivernale en milieu clos (Scandinavie, Islande, montagne).
. Les fermiers sont exposés notamment là où le travail auprès des animaux s’effectue pendant une longue période hivernale en milieu clos (Scandinavie, Islande, montagne).
On considère comme acariens de stockage les Lepidoglyphus, Tyrophagus, Acarus, Glycyphagus, Aleuroglyphus et Chortoglyphus. Souvent c’est Lepidoglyphus qui domine ![]()
![]()
![]()
![]() .
.
L’exposition aux acariens de stockage n’est pas exclusive d’une exposition aux Dermatophagoides (ou Blomia). Dans leur grande majorité, les patients positifs pour un acarien de stockage sont positifs aussi pour un acarien domestique ![]()
![]()
![]() .
.
Blomia tjibodas a été décrit dans des fermes en Allemagne ![]() et pourrait positiver des tests pour Blomia tropicalis.
et pourrait positiver des tests pour Blomia tropicalis.
Une double positivité, au moins in vitro, pour un acarien domestique et un acarien de stockage sera donc fréquente. Comment interpréter cette double positivité ?
- chez un patient professionnellement exposé, une double sensibilisation est probable, par exemple Dermatophagoides + Lepidoglyphus. Des réactivités croisées de Lepidoglyphus peuvent positiver d’autres acariens de stockage dont la présence dans l’environnement du patient n’est pas établie (ex. Glycyphagus et Tyrophagus).
- chez un patient a priori non exposé à des acariens de stockage, tel un habitant des villes, le bilan sera moins clair.
- On pensera d’abord à une positivité pour les acariens de stockage induite par l’acarien domestique. De fait, l’observation des réactions croisées montre que, dans ces conditions, Dermatophagoides inhibe aisément l’acarien de stockage mais que la réciproque n’a pas lieu : l’acarien sensibilisant est celui qui domine la réaction croisée, donc Dermatophagoides
 .
. - Le répertoire des IgE du patient a alors plus d’affinité pour Dermatophagoides que pour l’acarien de stockage. S’il faut traiter, c’est sur la sensibilisation vis à vis de Dermatophagoides qu’il faut jouer.
- On pensera d’abord à une positivité pour les acariens de stockage induite par l’acarien domestique. De fait, l’observation des réactions croisées montre que, dans ces conditions, Dermatophagoides inhibe aisément l’acarien de stockage mais que la réciproque n’a pas lieu : l’acarien sensibilisant est celui qui domine la réaction croisée, donc Dermatophagoides
- Ceci étant, l’absence totale d’acariens de stockage en milieu urbain est difficile à prouver (Ebner dans
 ).
). - Et les matelas des maisons en Islande ont beau être dépourvus d’acariens, 9% des Islandais ont été trouvés positifs en RAST pour D. pteronyssinus dans l’étude ECHRS
 . Dans ce cas, il a été formulé une hypothèse selon laquelle les patients se seraient sensibilisés au cours de leur jeunesse lors de camps à la ferme.
. Dans ce cas, il a été formulé une hypothèse selon laquelle les patients se seraient sensibilisés au cours de leur jeunesse lors de camps à la ferme. - Cela montre en tous cas qu’il est important de revenir sur les conditions d’exposition prévalant dans le passé du patient.
Enfin, une positivité in vitro pour un acarien inattendu pourrait provenir d’une réactivité croisée glucidique (cf. Acariens et CCD).
- Il est probable que cette éventualité est marginale, bien que pouvant expliquer en partie les fortes prévalences de RAST positifs pour des acariens de stockage relevées en population urbaine
 .
.
Acariens et « allergènes cachés »
Les acariens peuvent aussi provoquer des allergies alimentaires : c’est le « pancake syndrome » ![]()
![]() .
.
Des acariens s’étant multipliés dans/sur un aliment peuvent provoquer une réaction allergique (parfois sévère) à l’ingestion de ces aliments, même après cuisson, chez des patients ayant une allergie respiratoire aux acariens. De nombreux cas ont été publiés ![]()
![]()
![]()
![]()
![]()
![]() .
.
Dans la série d’observations de Sanchez-Borges 24 des 30 patients étaient des adultes ![]() .
.
On sait que les allergènes du groupe 2, notamment, sont thermostables ![]() . Et des TC positifs ont été trouvés avec des farines contaminées préalablement chauffées 1 heure à 100°C
. Et des TC positifs ont été trouvés avec des farines contaminées préalablement chauffées 1 heure à 100°C ![]() .
.
Dermatophagoides farinae porte un nom montrant bien que cet acarien ne se plait pas que dans les literies. La plupart des cas de « pancake syndrome » étaient dus à des farines contaminées par des acariens, mais il a aussi été rapporté des cas de contamination par des débris de blattes ![]()
![]() ou par des moisissures
ou par des moisissures ![]() .
.
D’autres acariens peuvent aboutir aux mêmes réactions alimentaires : Tyrophagus, Suidasia, Aleuroglyphus, Blomia freemani, D. pteronyssinus, Thyreophagus ...
En dehors de la farine de blé (pancakes, gâteaux, aliments panés, pizzas, etc) on a noté des contaminations dans la farine de maïs (pollenta), avec diverses graines, des fruits secs, des poissons séchés, voire du thé ![]() .
.
Une étude au Japon a montré que 5,5% des échantillons de farine recueillie dans des cuisines familiales étaient contaminées par des D. farinae ![]() . Cela ne se produisait pas si la farine était gardée au réfrigérateur plutôt que dans un placard. Et dans un travail mené au Venezuela, 37% des farines testées étaient contaminées
. Cela ne se produisait pas si la farine était gardée au réfrigérateur plutôt que dans un placard. Et dans un travail mené au Venezuela, 37% des farines testées étaient contaminées ![]() .
.
Le diagnostic repose sur un test cutané positif avec l’aliment incriminé et négatifs avec le même aliment non contaminé.
A noter qu’une anaphylaxie alimentaire induite par l’effort a été décrite avec une farine contaminée (par Suidasia) ![]() .
.
Et la fréquence très élevée d’une intolérance aux AINS relevée dans 2 larges séries de patients ![]()
![]() peut justifier la recherche de facteurs adjuvants.
peut justifier la recherche de facteurs adjuvants.
Diagnostic d’une sensibilisation aux acariens
Tests basés sur des extraits
A priori, les acariens ne posent pas de difficultés diagnostiques majeures. En effet, certains allergènes représentent une part notable de l’IgE-réactivité et les extraits sont plus ou moins standardisés sur ces allergènes.
Par exemple, Hales ![]() donne une part de 50% pour les groupes 1+2 dans l’IgE-réactivité totale, et 10% pour chacun des groupes 4, 5 et 7.
donne une part de 50% pour les groupes 1+2 dans l’IgE-réactivité totale, et 10% pour chacun des groupes 4, 5 et 7.
Pourtant la multiplicité des sortes d’allergènes, le fait que certains se retrouvent dans les fèces (groupes 1 et 3 par exemple) ou au contraire dans le corps de l’animal (tropomyosines, paramyosines, par exemple), le manque d’hydrosolubilité (ex. apolipophorines) et la fragilité de certains allergènes dans un milieu contenant de nombreuses protéases font que les extraits peuvent différer sensiblement les uns des autres suivant les techniques de culture, récolte et extraction des fabricants d’extraits ![]()
![]()
![]() . Ainsi, le groupe 7 serait sous-représenté dans les extraits
. Ainsi, le groupe 7 serait sous-représenté dans les extraits ![]() .
.
On a relevé des écarts entre les contenus en Der p1 pouvant aller d’un facteur 1 à 6 entre 9 extraits commerciaux, et même de 1 à 20 pour Der p 2 ![]() .
.
A cela s’ajoutent des variations issues de différences entre les acariens cultivés et ceux avec lesquels le patient est en contact.
- On a pu montrer, par exemple, la présence de lignées génétiques différentes de sorte que les Der f 2 d’acariens européens étaient différents des Der f 2 d’Asie
 , ou que l’isoforme Der p 2.0101 était absente des acariens recueillis à Bangkok
, ou que l’isoforme Der p 2.0101 était absente des acariens recueillis à Bangkok  .
.
On peut donc craindre des différences entre les concentrations (et la répartition) des allergènes effectivement inhalés par les patients et des allergènes présents dans les extraits pour le diagnostic ![]() .
.
Globalement, il est cependant admis que les extraits sont acceptables pour être utilisés en immunothérapie pour autant que le patient soit réactif à des allergènes du groupe 1 et/ou du groupe 2 ![]()
![]() .
.
Cela pose donc la question de pouvoir affiner le diagnostic à l’aide d’allergènes purifiés ou recombinants.
Ceci est abordé ci-après. Au préalable, il convient de noter :
- la piètre corrélation entre un résultat quantitatif (kU/l) pour un CAP acarien et l’expression clinique : on voit, par exemple, qu’une probabilité de 35% pour des sifflements chez un enfant correspond aussi bien à 3 kU/l qu’à 100 kU/l

- l’effet d’entraînement sur les kU/l pour D. pteronyssinus par l’élévation globale des IgE totales en cas d’eczéma atopique : le rapport moyen acarien/IgE totales est à peu près le même chez un asthmatique et chez un sujet porteur d’eczéma atopique

- le peu de travaux jusqu’à présent pour valider les tests d’activation des basophiles s’agissant des acariens

Tests utilisant les ressources de l’allergologie moléculaire
Peut-on remplacer les extraits par des allergènes recombinants ? La réponse n’est pas simple.
1 – Comment choisir les allergènes les plus pertinents ?
1A- sur la base d’une forte prévalence de positivité
De multiples études ont donné des taux de positivité pour tel allergène ou groupe d’allergènes. Il s’agissait, le plus souvent, d’études d’IgE-réactivité in vitro. Le tableau ci-dessous donne une idée de ces prévalences à partir d’une cinquantaine de travaux :
Ordres de grandeur des positivités in vitro (NB : les différences régionales peuvent être à l’origine de taux très variables ; les chiffres entre parenthèses signifient un nombre très restreint de travaux)
| Groupe | Dermatophagoides | Blomia |
|---|---|---|
| 1 | 60-100 % | 60-80 |
| 2 | 60-95 | 40-60 |
| 3 | 20 et >> | 50 |
| 4 | 25-75 | 10 et >> |
| 5 | 30-60 | 50-80 |
| 6 | 40-50 | 10-20 |
| 7 | 20-50 | |
| 8 | 10-50 | |
| 9 | 30 et >> | (25) |
| 10 | 10-80 | (30) |
| 11 | 50-70 | (10-50) |
| 12 | (20-50) | |
| 13 | (10-50) | |
| 14 | (40-70) | |
| 15 | (70) | |
| 16 | (30-40) | |
| 17 | (35) | |
| 18 | (60) | |
| 19 | (10-20) | |
| 20 | (10-30) | |
| 21 | (25) | (90) |
| 22 | (50) |
On peut constater que, contrairement à d’autres sources d’allergènes, la réactivité se répartit sur de nombreux allergènes pour les acariens, sans prédominance évidente pour une catégorie précise.
Les allergènes des groupes 1 et 2 sont souvent présentés comme les plus pertinents :
- dans une étude européenne multicentrique, une positivité pour Der p 1 et/ou Der p 2 était trouvée chez plus de 97% des patients

- de même, chez des patients dont la réactivité était maximale pour les groupes 4 ou 5 ou 7, il était malgré tout observé une positivité pour un allergène du groupe 1 et/ou du groupe 2

Mais des différences régionales existent, tant en Europe que d’un continent à un autre :
- les patients mono-réactifs à Der p 1 ou à Der p 2 sont beaucoup moins fréquents en France (18%) qu’en Italie, Suède ou Autriche

- la positivité pour les groupes 1 et 2 était respectivement de 85-100% et 63-96% dans cette étude européenne, mais de 76% et 74% en Australie
 et de 64% et 69% en Colombie
et de 64% et 69% en Colombie  , par exemple
, par exemple - des différences encore plus grandes étaient vues pour certains autres groupes d’allergènes : par exemple 74% de positivité en Australie contre 28-45% dans l’étude européenne
1B – sur la base du niveau de l’IgE-réactivité
Les prévalences de positivité ne sont pas parallèles aux niveaux d’IgE-réactivité de chacun des allergènes, comme le montre le graphique ci-dessous ![]()

Aussi, serait-il préférable de choisir comme pertinents les allergènes qui représentent la plus grande part de l’IgE-réactivité globale.
En dehors de l’étude de Hales schématisée ci-dessus, peu de travaux ont cherché à comparer les positivités et l’IgE-réactivité de façon quantitative pour la plupart des allergènes d’une même espèce d’acariens.
L’équipe de Chew a présenté des résultats pour D. farinae et Blomia tropicalis en étudiant une cohorte de patients Singapouriens (n=181) ![]() . Pour ces auteurs, il faut ajouter le groupe 13 au rang des allergènes les plus importants. Le tableau ci-dessous montre la part relative d’IgE-réactivité des 10 premiers allergènes parmi les 30 étudiés par Chew.
. Pour ces auteurs, il faut ajouter le groupe 13 au rang des allergènes les plus importants. Le tableau ci-dessous montre la part relative d’IgE-réactivité des 10 premiers allergènes parmi les 30 étudiés par Chew.
Part relative de l’IgE-réactivité de divers allergènes, rapportée à celle de l’allergène le plus IgE-réactif. Valeurs trouvées à l’aide de recombinants (Singapour)
| D. farinae | Blomia tropicalis | |||
|---|---|---|---|---|
| Groupe d’allergènes |
IgE-réactivité relative |
Groupe d’allergènes |
IgE-réactivité relative | |
| 1 | 1 | 5 | 1 | |
| 2 | 0,58 | 13 | 0,85 | |
| 5 | 0,45 | 1 | 0,51 | |
| 4 | 0,20 | 3 | 0,39 | |
| 13 | 0,19 | 12 | 0,25 | |
| 14 | 0,18 | 14 | 0,25 | |
| 3 | 0,18 | 2 | 0,19 | |
| 7 | 0,10 | 11 | 0,12 | |
| Etc … | Etc … | |||
2 – Vers un choix restreint d’allergènes ?
Plusieurs travaux ont cherché à savoir si l’association de 2 ou 3 allergènes avait une sensibilité diagnostique proche de 100% :
- en associant rDer p 1, rDer f 1 et rDer p 2, une sensibilité diagnostique de 94,8% a été obtenue chez des sujets Brésiliens, et même de 98,3% en y ajoutant 2 autres tests rBlo t 5 et rDer p 5

- l’association des groupes 1 et 2 de D. pteronyssinus et D. farinae détectait 82% des patients au Japon et 89% en Espagne
 . Le déficit par rapport à 100% n’était pas du au groupe 7 car tous les patients positifs pour Der p 7 l’étaient aussi pour le groupe 1 ou le 2
. Le déficit par rapport à 100% n’était pas du au groupe 7 car tous les patients positifs pour Der p 7 l’étaient aussi pour le groupe 1 ou le 2 - la même observation était présente dans la cohorte étudiée par Hales en Australie

- la sensibilité diagnostique dépassait 97% avec groupe 1 et/ou 2 dans l’étude de Weghofer

- elle était de 93% en Colombie avec Der p 1 + Der p 2 + Der p 10 (tropomyosine)

- et en tests cutanés une association de Der p 1, f1 , p 2 et p 5 montrait une sensibilité de 76%

Quelles sont les suggestions des auteurs ? :
- Arruda opte pour Der p 1 + p 2 + p 5 <biblio9291
- Thomas penche pour 1 + 2 + 4 + 5 + 7, et n’élimine pas les groupes 11, 14 ou 15 éventuellement

- Weghofer estime que les groupes 1 et 2 suffisent, même si l’ajout des groupes 5 et 7 serait intéressant
- enfin, il ne semble pas pertinent d’adjoindre une tropomyosine car la réactivité à cette famille d’allergènes est plutôt le résultat d’une sensibilisation tierce (ex. crustacés, helminthes)

Ces associations d’allergènes donnent des sensibilités diagnostiques très élevées. Il convient de remarquer cependant que ces taux sont artificiels : ils résultent de l’addition des positivités séparées de chacun des allergènes. Si les allergènes étaient mélangés dans le même test on obtiendrait une sensibilité moindre par simple effacement des combinaisons statistiques.
Pour le moment, la société Phadia ne propose pas encore de tests in vitro avec des recombinants d’acariens. Des tests sont disponibles avec la technique DPC-Siemens pour les allergènes naturels Der p 1 et Der p 2 de D. pteronyssinus.
Peut-on se satisfaire de l’IgE-réactivité aux groupes 1 et 2 des allergènes d’acariens ? :
- en histamino-libération, Der p 1 ne représente qu’une part réduite de l’activité suscitée par un extrait de D. pteronyssinus

- un extrait déplété en Der p 1 conserve quand même 70% de son activité

- Der p 1+Der p 2, ou Der f 1+Der f 2, ne représentent que 80 voire 60% de l’IgE-réactivité d’un extrait

- en challenge bronchique Der p 1 et Der p 2 donnent des réponses retardées plus faibles qu’un extrait de D. pteronyssinus

- et les recombinants montrent parfois une IgE-réactivité inférieure à celle de leurs homologues naturels : moins 50% pour rDer p 1 ou rDer f 1, moins 30% pour rDer p 2 et moins 20% pour rDer f 2
 . Ces déficits de réactivité se retrouvaient en histamine release.
. Ces déficits de réactivité se retrouvaient en histamine release.
Le choix entre allergène naturel et allergène recombinant se pose donc.
Un même allergène naturel est souvent un mélange d’isoformes et de variants, voire de paralogues ayant de grandes différences entre eux. Par exemple, Blo t 5 ![]() avec lequel il a été relevé des rBlo t 5 négatifs alors que les mêmes sujets avaient un nBlo t 5 positif
avec lequel il a été relevé des rBlo t 5 négatifs alors que les mêmes sujets avaient un nBlo t 5 positif ![]() .
.
Les recombinants sont-ils finalement trop sélectifs dans le cas des acariens ?
Améliorer les mesures in vitro ?
Il est classique d’observer des différences entre le résultat quantitatif (en kU/l) pour un extrait, disons du produit X, et la somme des résultats obtenus avec un panel d’allergènes naturels ou recombinants de ce même produit X. Par exemple, la somme des réactivités pour 9 recombinants de D. pteronyssinus varie de 50 à 300 KU/l pour une même réponse à 100 kU/l avec le CAP D. pteronyssinus global ![]() .
.
Ceci est-il du à un manque de standardisation des tests in vitro ? On sait qu’il n’y a pas interchangeabilité entre les unités des tests d’IgE « spécifiques », les kU/l, et celles pour les IgE totales, les UI/ml. Seules ces dernières sont adossées à un standard international OMS : une UI/ml équivaut à 2,4 ng/ml d’IgE.
Pour pallier cette difficulté, il a été proposé d’utiliser un mode de calibration des tests d’IgE « spécifiques » qui soit, lui aussi, adossé à l’étalon OMS. Un anticorps chimérique a été synthétisé à cet effet par recombinaison génétique à partir :
- de la partie Fc d’une IgE humaine
- de la partie Fab d’un anticorps monoclonal murin anti-Der p 2
Selon les promoteurs de cette technique ![]() , l’anticorps chimérique étant aisément quantifiable en UI/ml d’IgE totales (il possède une partie identique aux IgE humaines) peut servir pour calibrer un test in vitro, tel un CAP, et donc permettre de rendre des résultats d’IgE spécifiques pour des patients en UI/ml également.
, l’anticorps chimérique étant aisément quantifiable en UI/ml d’IgE totales (il possède une partie identique aux IgE humaines) peut servir pour calibrer un test in vitro, tel un CAP, et donc permettre de rendre des résultats d’IgE spécifiques pour des patients en UI/ml également.
Les résultats étant dorénavant standardisés en UI/ml, on pourrait espérer retrouver une meilleure adéquation entre un test global (ex. D pteronyssinus) et la somme des valeurs obtenues avec les allergènes du même produit.
Mais c’est oublier des fondamentaux ! :
- un anticorps monoclonal n’est pas représentatif de la diversité des épitopes reconnus par les patients, même sur un allergène unique, et a fortiori si l’anticorps est d’origine non humaine (ici souris)
- une calibration avec un « étalon » anti-Der p 2 aurait peut-être une utilité pour tester la réactivité de patients vis-à-vis de Der p 2 … mais pas pour Der p 1, etc, etc… Il faudrait créer autant d’étalons qu’il y a d’allergènes différents !!
- et quand ce principe est proposé pour s’appliquer non plus à des molécules définies, comme Der p 2, mais à un extrait comme D. pteronyssinus, on peut considérer que la tentative est vouée à l’échec !!
Quelques travaux ont été publiés s’appuyant sur ce type d’étalonnage. Il va de soi que les conclusions qui en sont tirées sont sujettes à caution ![]()
![]() , quand elles ne sont pas contraires à la science
, quand elles ne sont pas contraires à la science ![]() .
.
Acariens et CCD
(voir aussi : Les CCD)
On estime à 20-30% la proportion des sujets polliniques porteurs d’IgE contre des glyco-épitopes. Les patients allergiques aux acariens (et non polliniques) ont rarement un CAP broméline positif ![]()
![]() . On pourrait donc penser que les tests in vitro pour les acariens ne sont pas influencés par les CCD.
. On pourrait donc penser que les tests in vitro pour les acariens ne sont pas influencés par les CCD.
Divers résultats tendent à modérer cette conclusion :
- chez des sujets vus pour suspicion d’allergie respiratoire (n=4535) la prévalence de CAP positifs pour D. pteronyssinus passe de 3% en cas de CAP broméline négatif à 30% en cas de CAP broméline positif

- le periodate tend à diminuer l’IgE-réactivité d’un extrait de D. farinae
 ou de Lepidoglyphus
ou de Lepidoglyphus 
- chez un sujet avec des IgE anti-CCD, la broméline inhibe en partie le CAP D. pteronyssinus

- une partie de l’IgE-réactivité vis à vis de D. pteronyssinus ou D. farinae peut être capturée par la broméline et/ou la peroxydase de raifort (HRP)
 .
. - dans leur série de patients allergiques aux hyménoptères (n=259), Kochuyt et coll. relevaient 17% de CAP acariens positifs parmi les sujets CCD positifs, contre 4% parmi ceux qui étaient CCD négatifs
 .
.
Existe-t-il des allergènes glycosylés pour expliquer cette glyco-réactivité des acariens ?
De fait, certains allergènes sont glycosylés (mais il faut rappeler que d’autres glycoprotéines que des allergènes dans l’extrait peuvent être reconnues par les IgE) ![]() :
:
- le groupe 1 : Der p 1, Der f 1, etc..
 mais pas Blo t 1
mais pas Blo t 1 

- le groupe 4

- le groupe 7
 , sauf Lep d 7
, sauf Lep d 7 
- le groupe 11


- le groupe 15

- Der f 18

Ces glycoprotéines d’acariens pourraient générer des IgE reconnaissant des épitopes glucidiques. Mais ces épitopes glucidiques d’acariens peuvent-ils croiser avec ceux des plantes ou des venins d’hyménoptères ?
- Pour cela il faudrait que des motifs glucidiques communs existent, comme un xylose ou un fucose-1,3. On connaît très mal les chaînes glucidiques des protéines d’acariens. Altmann
 estime que les acariens n’ont pas de chaînes avec un fucose-1,3.
estime que les acariens n’ont pas de chaînes avec un fucose-1,3. - Les cellules dendritiques reconnaissant Der p 1 par le biais de « récepteurs à mannose »
 , on peut supposer la présence de galactose et de béta-NAG sur Der p 1
, on peut supposer la présence de galactose et de béta-NAG sur Der p 1  . Mais des résultats contradictoires ont été obtenus avec les lectines
. Mais des résultats contradictoires ont été obtenus avec les lectines 
 .
. - Par ailleurs, un rDer f 1 muté au niveau du site de glycosylation conserve une IgE-réactivité équivalente à celle de l’allergène naturel nDer f 1
 .
.
Au total, si l’IgE-réactivité glucidique des acariens est encore assez mal connue, les tests in vitro pour les acariens semblent peu ou pas du tout influencés par la présence d’IgE anti-CCD. Dans la grande majorité des cas, les tests in vitro suivent bien les résultats des tests cutanés et sont concordants avec la clinique.



